A study from researchers at Boston Children's Hospital has identified the molecule that enables immune cells to release interleukin-1 (IL-1), a key cytokine in the innate immune response. The work was published yesterday in Immunity.
"Most proteins have a secretion signal that causes them to leave the cell," says senior author Jonathan Kagan, Ph.D. "IL-1 doesn't have that signal. Many people have championed the idea that IL-1 is passively released from dead cells: you just die and dump everything outside."
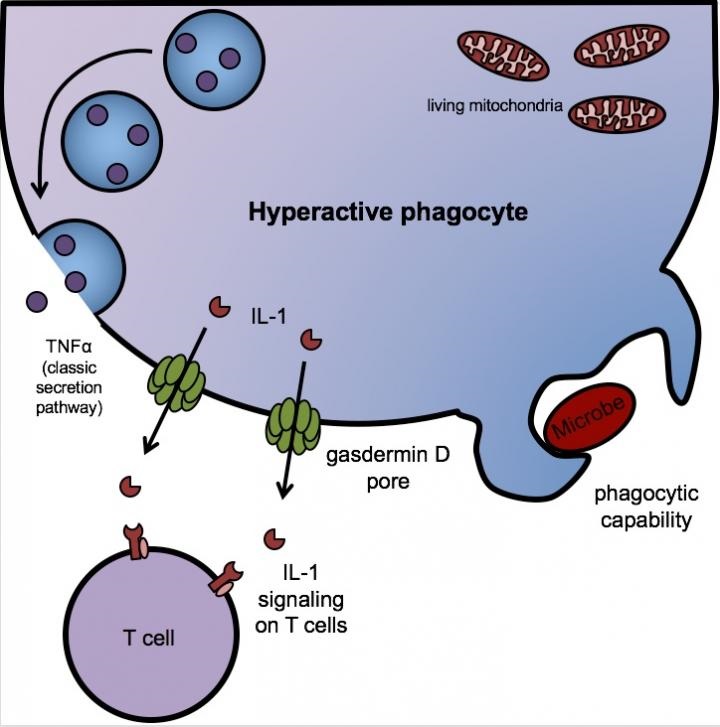
According to other published studies, a protein called gasdermin D punches holes in the membranes of infected cells by forming pores, leaving the cell to die. This causes the release of IL-1 and other cytokines.
In this new study, the researchers show that gasdermin D is also capable of triggering the release of IL-1 from living immune cells. The group's work involved hyperactive macrophages, immune cells that engulf and ingest pathogens. "We found, in hyperactivated cells, that gasdermin forms enough pores to let IL-1 pass through, but not enough to kill the cell," says Kagan.
 Kagan and the lab are now looking into targeting gasdermin D to stimulate IL-1 release from living, functional cells. They hope that their work will help create stronger and longer lasting vaccines.
Kagan and the lab are now looking into targeting gasdermin D to stimulate IL-1 release from living, functional cells. They hope that their work will help create stronger and longer lasting vaccines.
Image: In hyperactivated macrophages, gasdermin D forms a limited number of pores—enough to release IL-1 but not enough to kill the cell through pyroptosis. The macrophages stay alive and retain their phagocytic capacity, continuing to ingest microbes while stimulating memory T-cell formation via IL-1. Other proteins, like the cytokine TNF-alpha, are released through the classic secretion-signal pathway. Image courtesy of Charles Evavold, Boston Children's Hospital.